
Gives up 3 electrons, it gets a +3 charge. Up 2 electrons, it gets a +2 charge, and when it We call these "oxidation states." When iron gives Rust can come in different forms, because iron (symbol Fe) normally gives up 2 or 3 electrons when it forms compounds. (The oxygen you breathe is O 2, not elemental oxygen.) It is found all over everywhere iron and oxygen come in contact! Both iron and oxygen form compounds quite easily, so they are not readily found alone. It does not easily form compounds so it is found by itself, in stream beds (placer deposits) or in veins in rocks. Gold is also an element, in and of itself. The quick answer is: Diamond is a pure element, carbon gold is a pure element, gold and rust is a compound, Iron Oxide, of iron andĭiamond is pure elemental carbon, compressed to its crystal form, under extreme heat and pressure deep within the Earth. You can see things like steel rust because steel contains iron. Iron to iron oxide is an example of corrosion. Quickly with oxygen, and the process of turning 
Rust is the common name for iron oxide, which as the name implies, is made up of the elements iron and oxygen. However, this process takes thousands of years. Interestingly, graphite is the more stable form of carbon, so diamonds will eventually turn into graphite (diamonds aren't really forever!). The only difference between the two is the way that the atoms are bonded together. Also, graphite is also made up of carbon. The alloys are what you typically find in jewelry,Ĭarbon. Since pure gold is fairly soft, it is often combined with other metals to make alloys to make it a little stronger. Most rust has the chemical formula FeO 2H, also written as FeO (OH), and mineral name of this material is lepidocrocite (pronounced lay-pedo-kro-kite).įirst, gold is an element. Rust is usually a mixture of iron (symbol Fe) oxygen (symbol O) and some hydrogen (symbol H). In jewelry (which is the biggest use of gold) the gold is usually mixed with some copper (symbol Cu) Pure gold is usually only used to store money in banks. The element symbol Au comes from the Latin word for gold which is aurum. 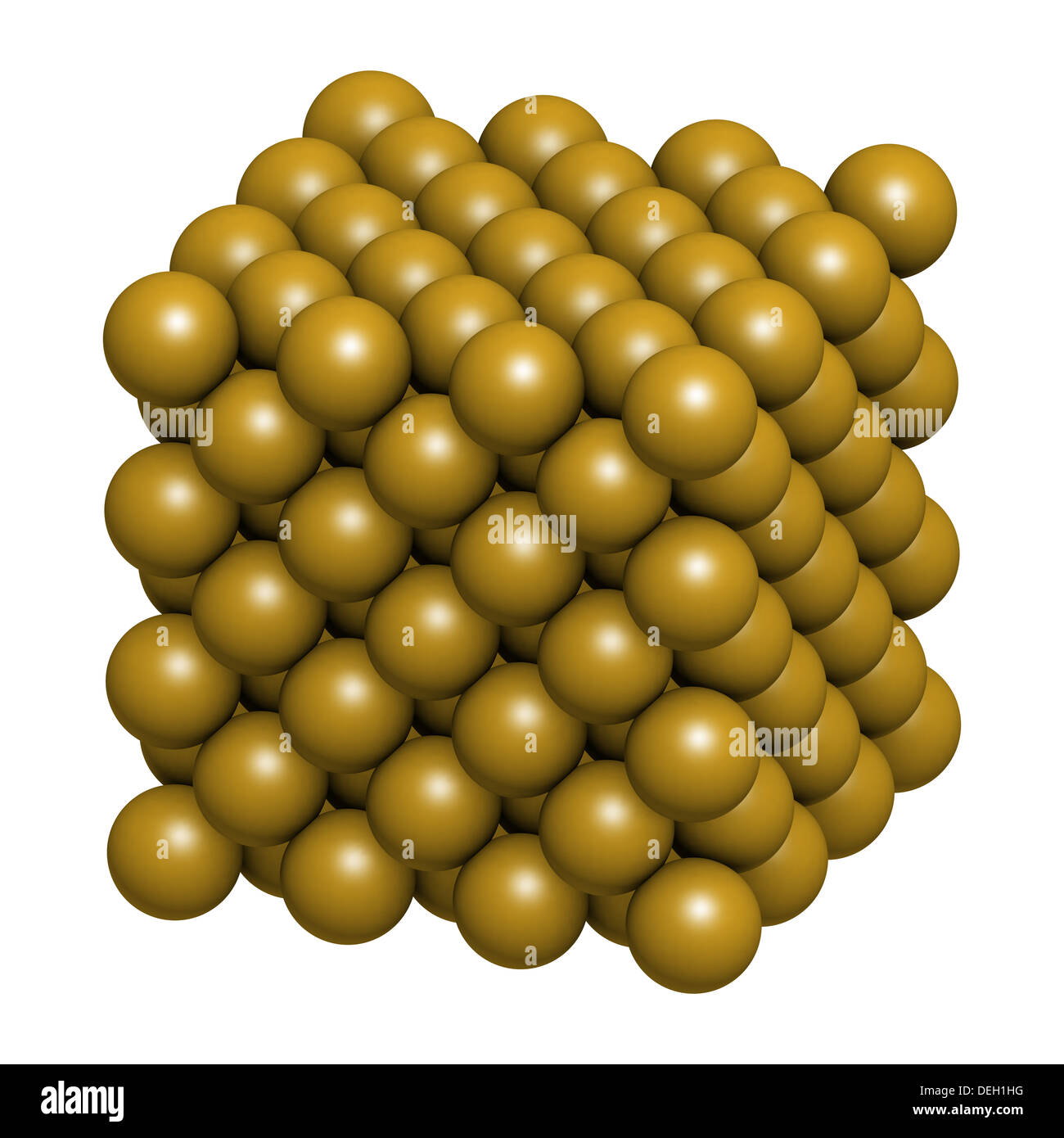
These tend to give diamonds different colors. Sometimes however, small amounts of carbons neighbors in the periodic table, such as boron (symbol B), and Nitrogen (symbol N) are also found in diamond. Diamonds, particularly high quality ones, are very pure carbon. The element in diamond is carbon (symbol C). That is, if you think you have some hematite, rub the mineral on an unglazed ceramic tile (called a streak plate) and the streak is blood red. Rust on the other hand is ferric oxide or Fe 2O 3. Gold, symbol Au is atomic number 79 and is like diamond, a pure substance. What elements are in diamonds, gold, and rust?ĭiamond is 100 % Carbon, atomic number 6 on the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
